 This is useful because it allows you to predict how a particular metal will undergo a certain chemical reaction. Metals are listed in the Reactivity Series from the most reactive to the least reactive. Today, we shall take a look at The Reactivity Series of Metals. In the previous blog post, we have discussed on the physical properties of pure metals as well as the four main reasons why we use metals in the form of alloys. IP Chemistry: Reaction of Metals with Water, Steam and Dilute Acids. You can watch the YouTube Video below to have a greater understanding of the reactions between metals and water, steam as well as dilute acids.Ĭlick on the following link for the video on O-Level Chemistry. YouTube Video Tutorial on Reaction of Metals with Water, Steam and Dilute Acids Thus, based on the reactions of metals with cold water, steam and dilute hydrochloric acid, we can place metals in order of their reactivity i.e. Lead does not appear to react with sulfuric acid because of the insoluble layer of lead (II) sulfate coated onto the underlying lead metal. Similarly, the above applies when sulfuric acid is being used. The chemical equation with state symbols for the reaction: This is being heavily covered in my Sec 3 GCE O-Level and IP Pure Chemistry Tuition Classes. Hence, lead does not appear to react with hydrochloric acid. As such, reaction slows down and eventually stops. 
This becomes a protective layer and prevents further reaction of the hydrochloric acid with the underlying lead metal. However, do note that when hydrochloric acid is being used, the initial reaction between lead and hydrochloric acid will form an insoluble layer of lead (II) chloride. 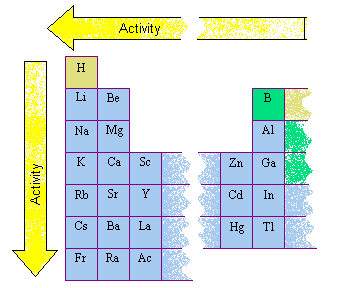
Note that lead should react easily with dilute acids since it is higher than hydrogen in the Reactivity Series. Sn(s) + 2HCl(aq) → SnCl 2(aq) + H 2(g) reacts slowly Zn(s) + 2HCl(aq) → ZnCl 2(aq) + H 2(g) reacts moderatelyįe(s) + 2HCl(aq) → FeCl 2(aq) + H 2(g) reacts slowly Mg(s) + 2HCl(aq) → MgCl 2(aq) + H 2(g) reacts rapidlyĢAl(s) + 6HCl(aq) → 2AlCl 3(aq) + 3H 2(g) reacts rapidly MetalĢK(s) + 2HCl(aq) → 2KCl(aq) + H 2(g) reacts explosivelyĢNa(s) + 2HCl(aq) → 2NaCl(aq) + H 2(g) reacts explosivelyĬa(s) + 2HCl(aq) → CaCl 2(aq) + H 2(g) reacts violently The reactions of the metals with the dilute acids will also indicate how reactive the metals are and this is used to place them in the Reactivity Series.Ī more reactive metal will react more violently with the dilute acid.Ĭheck out the table below on the observations and chemical equation for the reaction of metals with dilute acid. Metal + Dilute Acid → Salt Solution + Hydrogen Gas Most metals react with dilute acids to form a salt solution and hydrogen gas. Sn(s) + H 2O(g) → SnO(s) + H 2(g) reacts readilyī) Reaction of Metals with Dilute Hydrochloric Acid Zn(s) + H 2O(g) → ZnO(s) + H 2(g) reacts readilyģFe(s) + 4H 2O(g) → Fe 3O 4(s) + 4H 2(g) reacts slowly Mg(s) + H 2O(g) → MgO(s) + H 2(g) violent reactionĢAl(s) + 3H 2O(g) → Al 2O 3(s) + 3H 2(g) reacts readily Mg(s) + 2H 2O(l) → Mg(OH) 2(aq) + H 2(g) very slow reaction MetalĢK(s) + 2H 2O(l) → 2KOH(aq) + H 2(g) violent reactionĢNa(s) + 2H 2O(l) → 2NaOH(aq) + H 2(g) violent reactionĬa(s) + 2H 2O(l) → Ca(OH) 2(aq) + H 2(g) reacts readily Note that a more reactive metal reacts violently with steam.Ĭheck out the table below on the observations and chemical equation for the reaction of metals with cold water and/or steam. Metal + Steam → Metal Oxide + Hydrogen Gas Such metals will react with steam to form metal oxide and hydrogen gas. 
Some metals such as zinc and iron, do not react with cold water but they do react with steam. Note that a more reactive metal will react more violently with cold water. Metal + Water → Metal Hydroxide + Hydrogen Gas This reaction can be easily represented by the following word equation: The more reactive metals tend to react with cold water to form metal hydroxide (alkaline solution) and hydrogen gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
